Blogs
Blogs
Bot Image On Trial Site News
Trial Site News features the latest coverage of clinical research. Read the article here:
ProstatID Passes FDA 510(k) Administrative Review
The Bot Image team has been hard at work trying to obtain FDA clearance. We’re excited to announce that we have passed the first major milestone on July 21, 2020.
Successful Beta Test
We’re proud to announce that in partnership with our beta testing site, we processed our first test case using the cloud-based architecture on 6/18/2020.
A Conversation With Our CEO Dr. Randall Jones, PhD, MBA
We recently sat down with our CEO in his library to discuss the mission, vision, and values of Bot Image. We discussed ProstatID and the…
10 Questions With Our Founder
We recently sat down with our founder to ask 10 questions about the company, the industry, and what are the options men have when facing…
Saving Lives, Improving Prostate Cancer Detection with Artificial Intelligence
A prostate cancer diagnosis can be devastating news for the one in seven men who develop the disease in their lifetimes. Annually, 30,000…
Bot Image highlights performance of ProstatID in study
AI medical device company Bot Image is highlighting the performance of its ProstatID in a study evaluating its ability to detect, diagnose, and screen for prostate cancer.
The Use Of Prostatid In Cancer Detection, Diagnosis And Screening
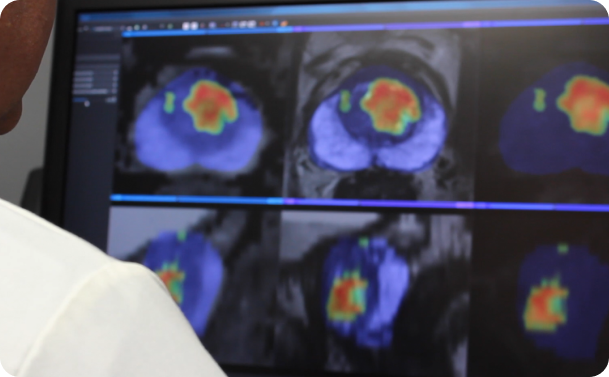
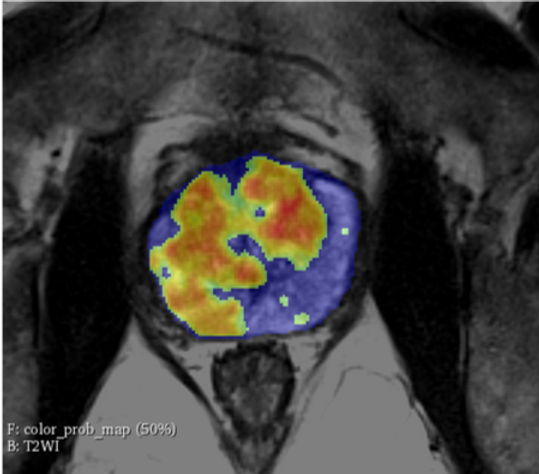
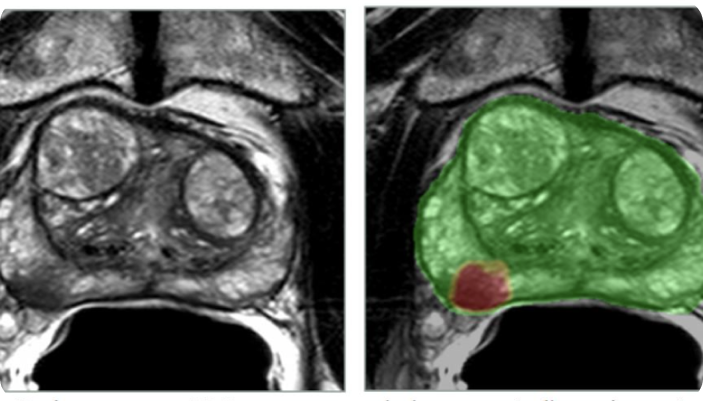
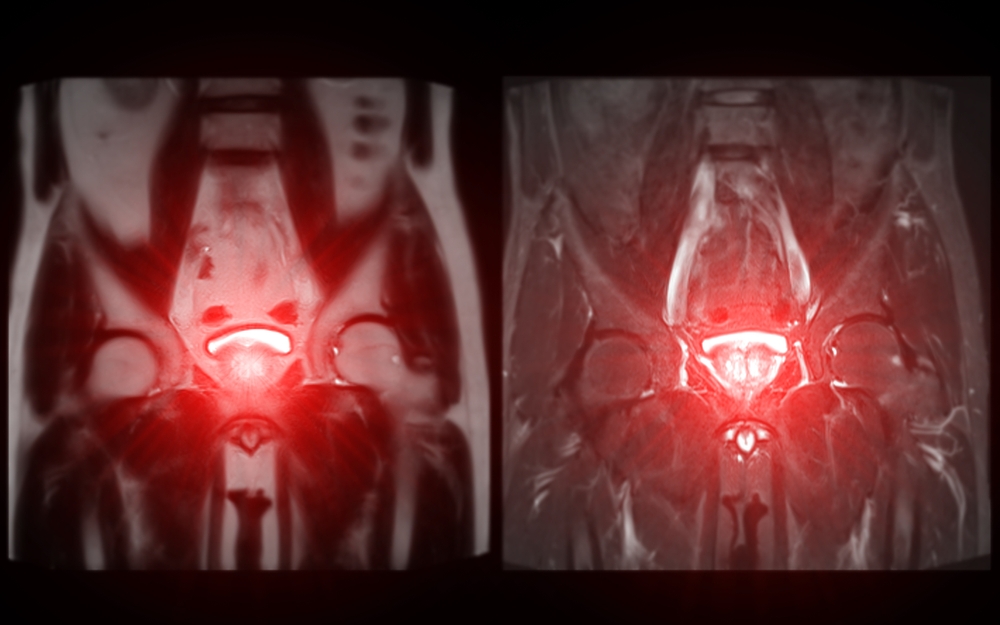
Overview: ProstatIDTM is North America’s first FDA-Cleared Prostate Cancer SCREENING, Detection and Diagnostic AI software. With 94% AUROC (Area under
An Overview of Prostate Cancer Detection Standard of Care and The Role of MRI for Early Detection of Prostate Cancer
Randall W. Jones, D.Eng. (PhD, MBA) EARLY DETECTION through Screening Population-Based Screening Studies Screening for Prostate Cancer (PCa) continues to
Are Prostate Cancer Screening Tests Accurate? The Truth You Need to Know
Introduction Prostate cancer is a significant concern for men, especially as they age. Early detection is crucial for effective treatment
When Should Men Get Screened for Prostate Cancer? A Comprehensive Guide
Prostate cancer is one of the most common cancers affecting men worldwide. However, despite its prevalence, there is still considerable
Decode the Subtle Signs of Prostate Cancer
Unveiling the Silent Threat Prostate cancer is a topic that many shy away from, yet understanding its early warning signs
Navigating Prostate Cancer Screening To Protect Your Health
Prostate cancer is one of the most common cancers among men worldwide, making early detection vital for effective treatment. Understanding
Screening Tests for Prostate Cancer What You Need to Know
Introduction Prostate cancer is a significant health concern for men worldwide. Early detection is crucial for successful treatment and better
When Should Men Start Screening for Prostate Cancer? Insights for Every Risk Level
Prostate cancer is one of the most common cancers affecting men worldwide. Early detection plays a crucial role in managing
Understanding Prostate Cancer Risk Factors to Protect Your Health
Introduction Prostate cancer is a significant health concern for many men worldwide. It is the second most common cancer among
Cracking the Code of Prostate Cancer Staging and Aggressiveness
Understanding how doctors determine the stage and aggressiveness of prostate cancer is crucial for patients and their loved ones. This
The Future of Prostate Cancer Diagnosis with AI-Powered MRI
Prostate cancer is one of the most common forms of cancer in men, and early detection is critical to improving
How AI is Revolutionizing Prostate Cancer Detection and Diagnosis
Most prostate cancers are either detected too late or over-diagnosed too early. For decades, the diagnostic pathway has been a
Why Non-Contrast MRI Is the Future of Prostate Cancer Screening
For years, contrast-enhanced MRI has been a cornerstone of prostate cancer diagnostics. While powerful, this approach is not without its
From Image to Insight: How ProstatID™ Fits Seamlessly Into Your Workflow
Adopting artificial intelligence in radiology should be straightforward, yet many systems introduce frustrating complexity. New workstations, manual uploads, and complicated
The Numbers Behind the FDA Clearance: Understanding ProstatID’s 93.6% AUROC
Every artificial intelligence company claims to offer “high accuracy.” It has become a baseline marketing promise in a crowded field.
Case Studies That Changed Minds: Real-World Results with ProstatID™
Data is one thing—clinical impact is another. In medical imaging, performance metrics and accuracy statistics are essential, but they only
AI Support for Radiologists: Reducing Variability and Improving Confidence
When experience varies, outcomes do too. This is a fundamental challenge in diagnostic medicine, and nowhere is it more apparent
The Economic Impact of Early Detection: Saving Billions with ProstatID™
The “wait and see” approach to medicine carries a hidden but staggering financial weight, particularly in oncology. For prostate cancer,
Why System-Agnostic AI Matters: Consistent Results Across Any MRI Platform
Many imaging centers hesitate to adopt artificial intelligence, and for good reason. The fear of vendor lock-in, complex integrations, and
Beyond Detection: How ProstatID™ Aids in Treatment Planning
In prostate cancer care, detection is only the first step. Many AI tools can flag a suspicious area on an
Building the Next Generation of Cancer Detection: The Future of Bot Image™ AI
A breakthrough in one area of medicine often serves as the blueprint for innovation in many others. With ProstatID™, we