Blogs, Articles & News
Blogs & News
A diagnosis journey often begins with uncertainty. For men facing the possibility of prostate cancer, that uncertainty has traditionally been
We are currently witnessing a renaissance in medical imaging. The first wave of Artificial Intelligence (AI) in healthcare focused heavily
In the rapidly evolving world of medical technology, “innovation” is a buzzword we hear daily. Startups and tech giants alike
Geography should not determine destiny. Yet, for millions of men living in rural or underserved communities, their zip code often
Medical innovation moves at the speed of light. Every day, researchers and engineers develop new algorithms, refine imaging techniques, and
The trajectory of modern medicine is shifting. For decades, the focus was often on volume—more tests, more procedures, and more
In the exclusive world of concierge medicine, “standard of care” is a starting point, not the goal. Patients who opt
In the rapidly evolving landscape of healthcare, specialization is the new standard of distinction. General hospitals and imaging centers are
The relationship between Primary Care Physicians (PCPs) and radiologists is one of the most critical axes in modern healthcare. It
The landscape of modern medicine is shifting beneath our feet. For decades, the practice of urology relied on a combination
In the highly competitive landscape of medical imaging, the difference between thriving and merely surviving often comes down to one
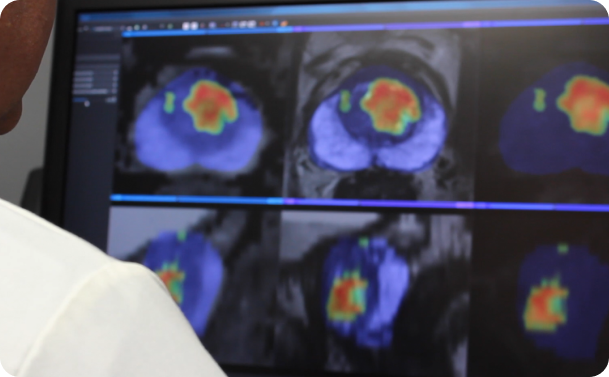
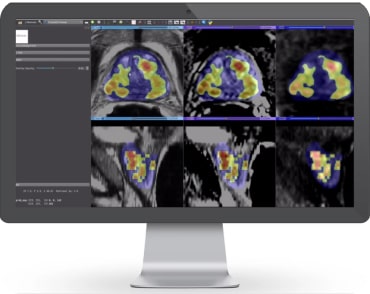
The integration of artificial intelligence into healthcare is unlocking unprecedented advancements in diagnostic medicine. AI-driven platforms can analyze medical images
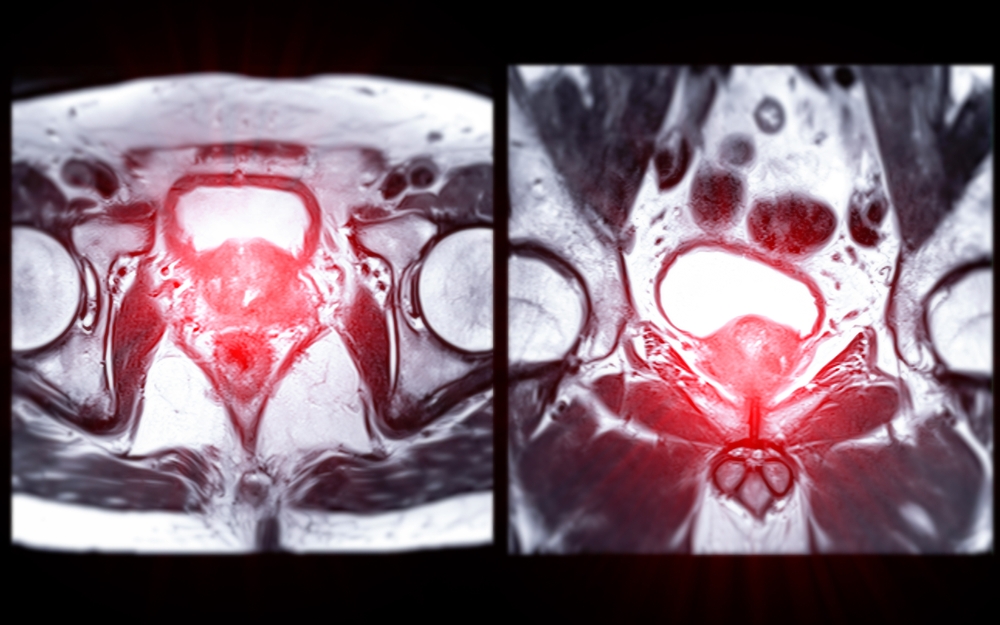
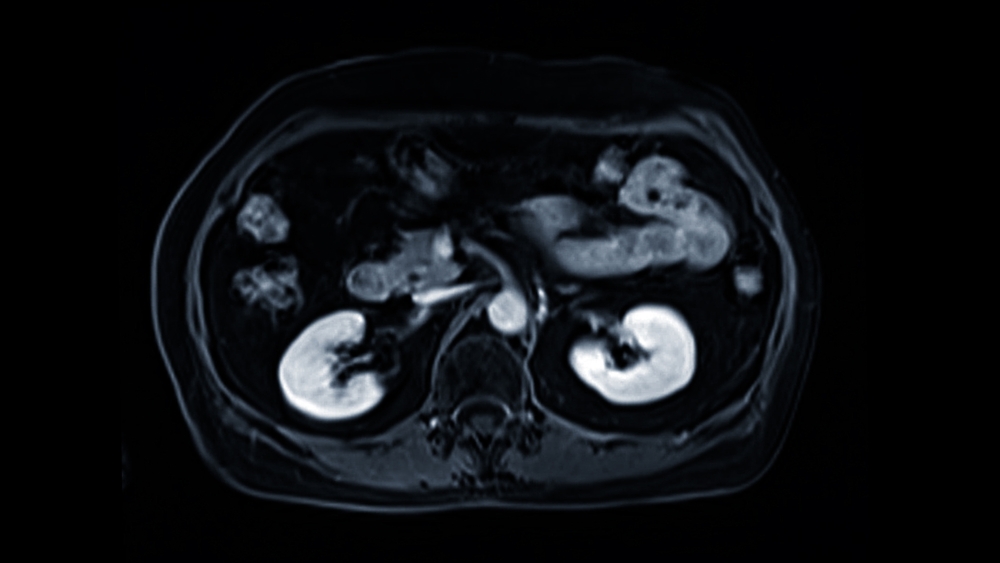
Magnetic Resonance Imaging (MRI) is one of the most powerful diagnostic tools in modern medicine. It provides incredibly detailed, non-invasive
The pathology report is a cornerstone of modern medicine. When a patient undergoes a prostate biopsy, the tissue samples are
Precision oncology represents a fundamental shift in how we approach cancer treatment. Instead of a one-size-fits-all strategy, it focuses on
The diagnosis of prostate cancer has long relied on a combination of clinical examination, PSA blood tests, and imaging. A
In the world of medical diagnostics, imaging and pathology have long been two distinct but complementary pillars. A radiologist interprets
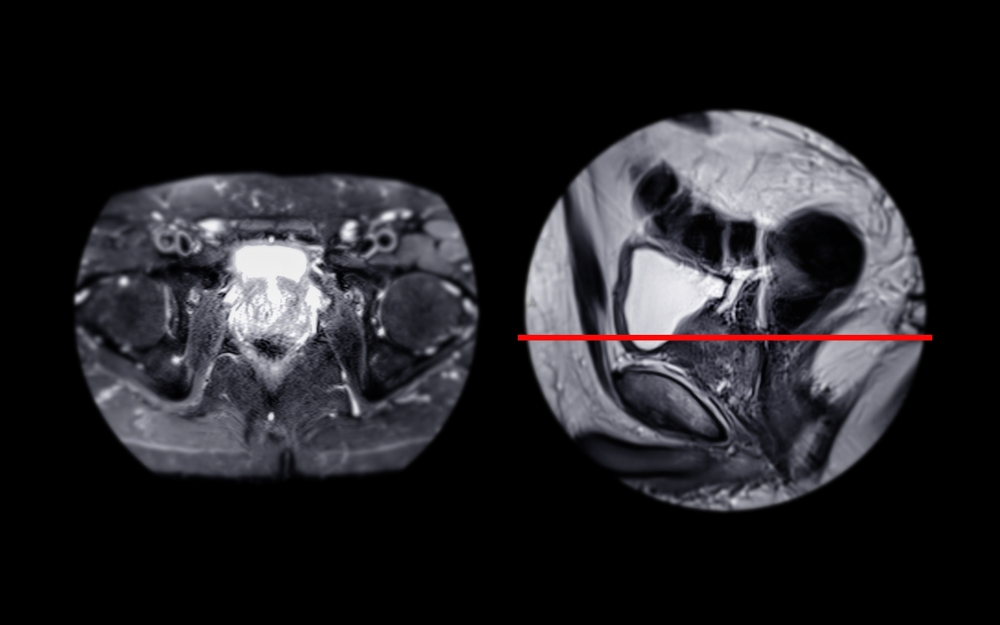
Medical imaging has taken remarkable leaps, moving from grainy, two-dimensional images to detailed, multi-layered scans. In prostate cancer diagnostics, Magnetic
In medical diagnostics, few outcomes are as emotionally charged or clinically significant as a false positive or a false negative.
Artificial intelligence is rapidly changing the landscape of medical diagnostics. AI-powered tools promise to deliver faster, more accurate, and more
The intersection of artificial intelligence and medical imaging is transforming diagnostics, offering new levels of precision and efficiency. Nowhere is
Hearing that you need a prostate MRI can bring up a lot of questions and concerns. When the term “AI-enhanced”
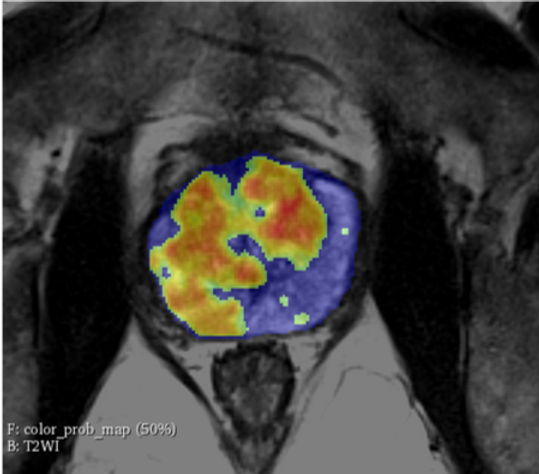
Artificial intelligence (AI) is revolutionizing prostate cancer diagnostics, offering a new level of precision in interpreting MRI scans. Powerful tools
The approach to managing prostate cancer is undergoing a profound transformation. For decades, the guiding principle was simple: find cancer
A potential cancer diagnosis is one of the most stressful experiences a person can face. The period of uncertainty between
The world of medical diagnostics is rapidly changing, and artificial intelligence (AI) is at the forefront of this transformation. For
The period between a medical test and its result can be one of the most stressful experiences in a person’s
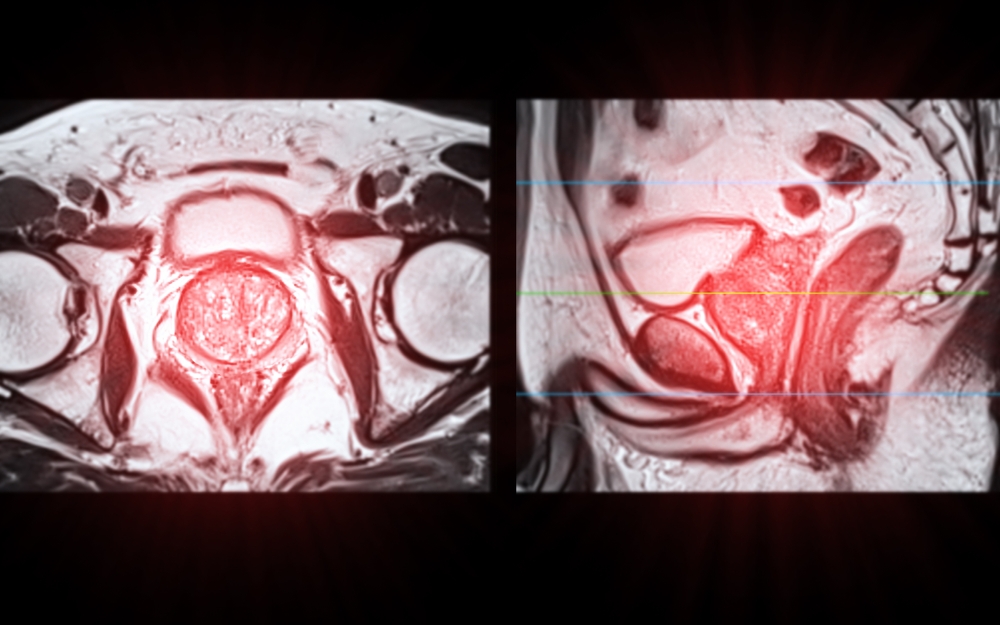
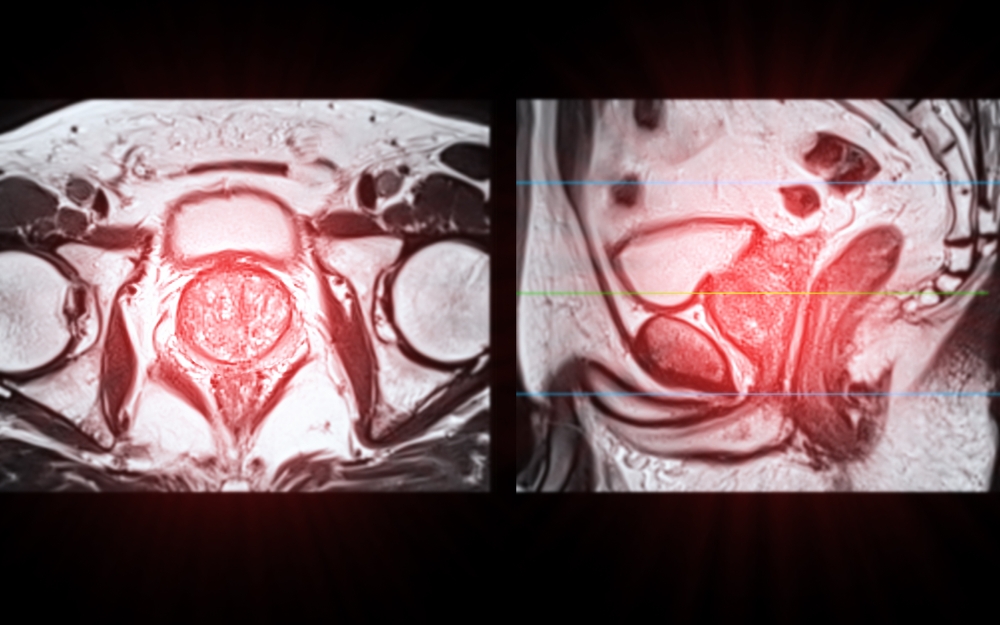
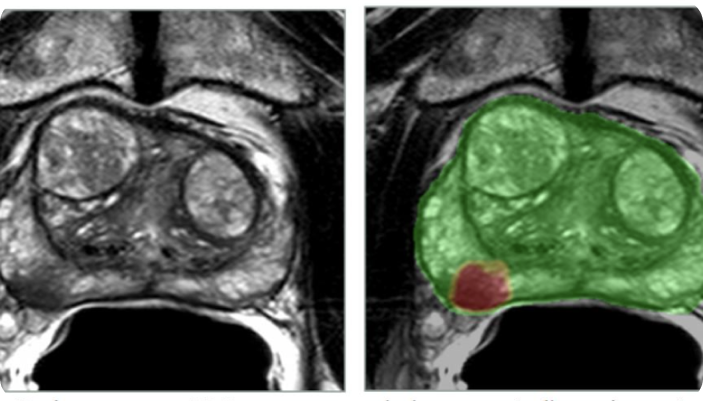
For decades, medical imaging has been presented in flat, two-dimensional slices. Radiologists and surgeons have been trained to mentally reconstruct
The words “MRI scan” can often bring to mind a long, noisy, and sometimes claustrophobic experience. For men on a
Navigating the world of prostate cancer screening can feel overwhelming. With a variety of tests, acronyms, and evolving technologies, it’s
In any medical imaging department, the MRI scanner is both a powerful diagnostic tool and a significant operational bottleneck. Patient
The decision to adopt new medical technology often hinges on two critical factors: clinical efficacy and financial viability. While the
Adopting artificial intelligence in a clinical setting can feel like a monumental task, often perceived as a complex and disruptive
In the competitive landscape of medical imaging, attracting a steady stream of referrals is the lifeblood of any successful center.
The diagnostic pathway for prostate cancer has long been a source of clinical uncertainty. For primary care physicians (PCPs), an
The modern radiology reading room is a paradox. It is a place of incredible technological sophistication, where high-definition monitors display
In the traditional timeline of medical imaging, “waiting” is the operative word. Patients wait for appointments. They wait in the
In the fast-evolving landscape of medical technology, “innovation” is a buzzword that gets thrown around daily. Vendors promise that their
Radiology is at a tipping point. The demand for diagnostic imaging is skyrocketing, driven by an aging population and a
In the high-stakes world of medical imaging, efficiency isn’t just a metric—it’s the lifeline of a successful practice. Imaging centers
In the high-stakes environment of cancer diagnosis, uncertainty is the enemy. A urologist looking at a prostate MRI isn’t just
Medical imaging is often compared to photography, but in the case of prostate cancer, it is more like solving a
Prostate cancer is a complex disease, but not all diagnoses carry the same weight. Some forms of the disease are
The narrative of “Man vs. Machine” has permeated almost every industry, from manufacturing to creative writing. In healthcare, and specifically
In the world of oncology, clinical guidelines are the bedrock of patient care. They provide physicians with evidence-based pathways for
The evolution of medical imaging is a story of constant refinement, driven by the pursuit of greater accuracy, improved patient
One of the most pressing challenges in modern urology is the prevalence of unnecessary prostate biopsies. For decades, the standard
The interpretation of prostate MRI is one of the most challenging tasks in modern radiology. It demands a high level
The prostate-specific antigen (PSA) test has long been the cornerstone of prostate cancer screening. For decades, it has served as
The landscape of prostate cancer diagnostics is undergoing a seismic shift, driven by the integration of artificial intelligence. For decades,
A breakthrough in one area of medicine often serves as the blueprint for innovation in many others. With ProstatID™, we
In prostate cancer care, detection is only the first step. Many AI tools can flag a suspicious area on an
Many imaging centers hesitate to adopt artificial intelligence, and for good reason. The fear of vendor lock-in, complex integrations, and
The “wait and see” approach to medicine carries a hidden but staggering financial weight, particularly in oncology. For prostate cancer,
When experience varies, outcomes do too. This is a fundamental challenge in diagnostic medicine, and nowhere is it more apparent
Data is one thing—clinical impact is another. In medical imaging, performance metrics and accuracy statistics are essential, but they only
Every artificial intelligence company claims to offer “high accuracy.” It has become a baseline marketing promise in a crowded field.
Adopting artificial intelligence in radiology should be straightforward, yet many systems introduce frustrating complexity. New workstations, manual uploads, and complicated
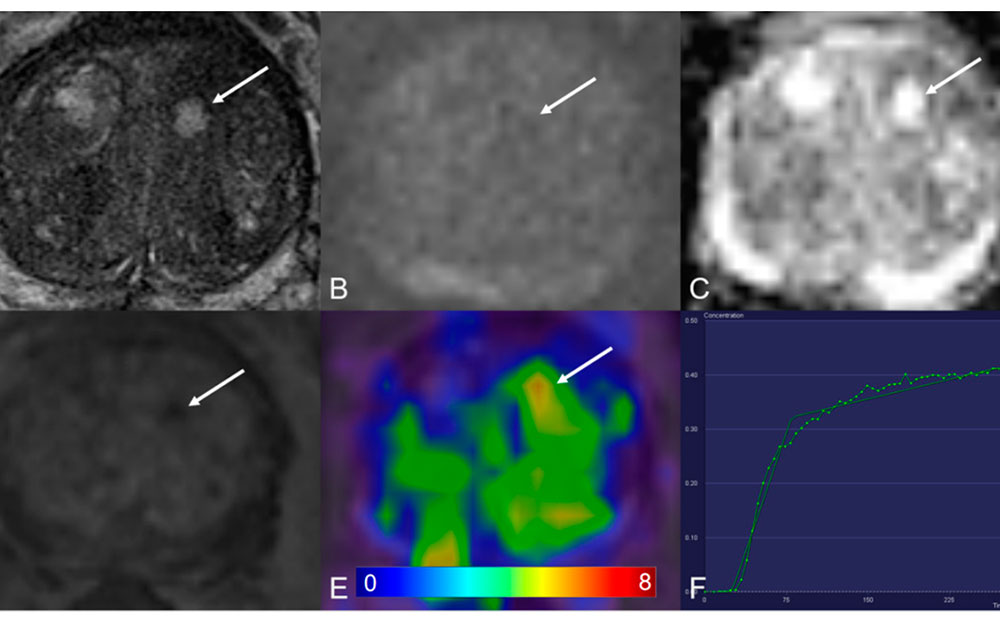
For years, contrast-enhanced MRI has been a cornerstone of prostate cancer diagnostics. While powerful, this approach is not without its
Most prostate cancers are either detected too late or over-diagnosed too early. For decades, the diagnostic pathway has been a
AI for prostate MRI classification has evolved rapidly, but data privacy, limited annotations, and domain variability still slow its translation
As artificial intelligence becomes central to prostate MRI interpretation, one fundamental question remains: can clinicians truly trust what they don’t
Artificial intelligence in prostate MRI is only as strong as the data it learns from. An AI model can learn
Despite major advances in artificial intelligence for prostate MRI, several real-world challenges remain. From subtle, low-contrast lesions and patient motion
As artificial intelligence continues to transform prostate MRI analysis, its success in everyday clinical practice hinges on more than just
AI-based prostate MRI lesion classification delivers tremendous diagnostic power, but its true impact depends on seamless integration into clinical workflows
The field of medical imaging is in a constant state of evolution, and nowhere is this more apparent than in
MRI-based prostate cancer lesion classification has moved beyond simple image interpretation. Today, it directly influences how physicians make critical diagnostic
Artificial intelligence in prostate MRI has made enormous strides. Research has shown that AI can identify, segment, and classify suspicious
Comparing artificial intelligence (AI) models across different studies is essential to understanding progress in prostate MRI lesion classification. Despite an
Developing artificial intelligence for prostate MRI isn’t just about training a model that performs well on paper. An algorithm can
When evaluating artificial intelligence for prostate MRI analysis, it’s tempting to ask for a single, simple “accuracy” score. However, in
Artificial intelligence (AI) in prostate MRI is transforming how we detect and diagnose cancer. However, one of the biggest challenges
Combining MRI-derived radiomic features with clinical data like PSA levels, biopsy results, and patient demographics is advancing prostate cancer lesion
Deep learning has transformed prostate MRI analysis by automating lesion detection, segmentation, and classification. These AI models, built on Convolutional
While deep learning architectures like neural networks dominate today’s AI headlines, classical machine learning (ML) algorithms remain highly relevant and
For radiomics to become a truly useful tool in clinical practice, its features must be consistent and comparable across different
In medical imaging, particularly for prostate MRI, how we teach a computer to “see” is everything. The goal is to
Radiomics is a powerful field that converts medical images, like a prostate MRI, into a vast amount of quantitative data.
Radiomics is the process of converting medical images, such as MRIs, into a vast amount of high-dimensional, quantitative data. This
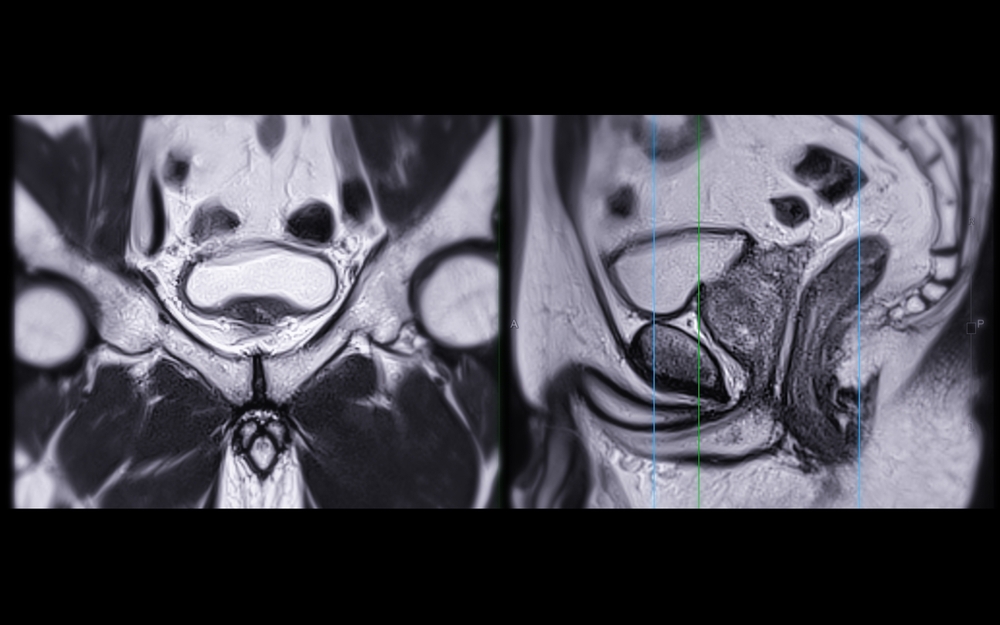
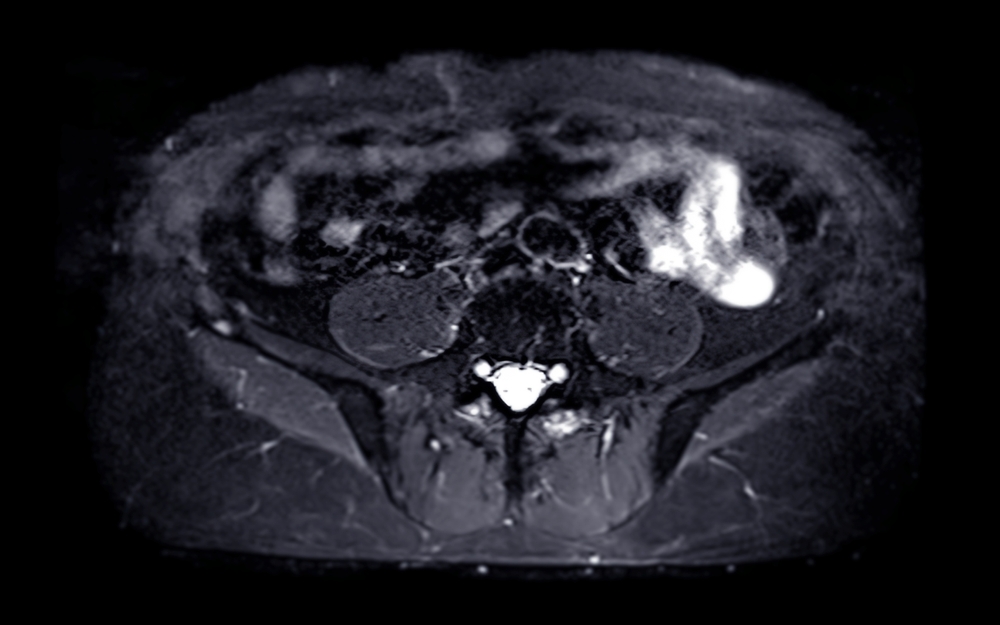
Prostate MRI is a powerful diagnostic tool, offering a detailed view inside the body to help physicians find and assess
Multi-parametric MRI (mpMRI) has become a cornerstone in the modern diagnosis and management of prostate cancer. By combining anatomical and
Dynamic Contrast-Enhanced MRI (DCE-MRI) is a powerful imaging sequence that adds a functional dimension to prostate cancer diagnosis. While T2-weighted
Magnetic Resonance Imaging (MRI) has transformed our ability to see inside the human body without invasive procedures. For prostate cancer,
Prostate cancer is one of the most common cancers affecting men, but early detection dramatically improves outcomes. For decades, clinicians
Can Young Men Get Prostate Cancer? Understanding the Facts Prostate cancer is often seen as a disease of older men,
Prostate cancer diagnosis is getting smarter, faster, and more precise. That’s the promise of AI and prostate cancer research coming
Expanding the Role of Deep Learning in Cancer Detection Deep learning, a powerful subset of artificial intelligence (AI), has fundamentally
Advancements in artificial intelligence (AI) are revolutionizing the field of cancer research, and Bot Image stands at the forefront of
Prostate cancer is one of the most common forms of cancer in men, and early detection is critical to improving
Understanding how doctors determine the stage and aggressiveness of prostate cancer is crucial for patients and their loved ones. This
Introduction Prostate cancer is a significant health concern for many men worldwide. It is the second most common cancer among
Prostate cancer is one of the most common cancers affecting men worldwide. Early detection plays a crucial role in managing
Introduction Prostate cancer is a significant health concern for men worldwide. Early detection is crucial for successful treatment and better
Prostate cancer is one of the most common cancers among men worldwide, making early detection vital for effective treatment. Understanding
Unveiling the Silent Threat Prostate cancer is a topic that many shy away from, yet understanding its early warning signs
Prostate cancer is one of the most common cancers affecting men worldwide. However, despite its prevalence, there is still considerable
Introduction Prostate cancer is a significant concern for men, especially as they age. Early detection is crucial for effective treatment
Randall W. Jones, D.Eng. (PhD, MBA) EARLY DETECTION through Screening Population-Based Screening Studies Screening for Prostate Cancer (PCa) continues to
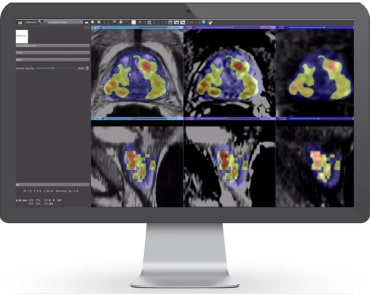
Overview: ProstatIDTM is North America’s first FDA-Cleared Prostate Cancer SCREENING, Detection and Diagnostic AI software. With 94% AUROC (Area under
TeraRecon, on March 18, 2023 CAMBRIDGE, Mass., Mar. 18,2023 /PRNewswire/ — The Eureka Clinical AI platform, from ConcertAI’s TeraRecon, now
FOR IMMEDIATE RELEASE Bot Image, an Omaha-based MRI medical device company has developed an AI-driven medical device CAD software to…
Just released, The Healthcare Disruptor, by Dr. Randall W Jones. Dr. Jones writes about his journey from rural cowboy to medical device expert and CEO challenging the healthcare system with his inventions…
Bot Image Inc: Facilitating Early Detection through Disruptive Technology In undetermined and unsettled times, one must be open to…
DR. RANDALL W. JONES FOUNDER & CEO OF BOT IMAGE, INC From joining the military at a young age to excelling at civilian jobs to…
Bot Image, Inc., a Nebraska and Maine based Artificial Intelligence medical device company, has received FDA-clearance on its ProstatIDTM AI
ProstatID, THE AI software FDA-cleared for not only detection and diagnosis of PCa, but also for screening using bpMRI. Yes,
Bot Image, Inc., a Nebraska and Maine based Artificial Intelligence medical device company (www.botimageai.com), raised the performance bar with its
AI medical device company Bot Image is highlighting the performance of its ProstatID in a study evaluating its ability to detect, diagnose, and screen for prostate cancer.
Dr. Randall Jones, CEO of BotImage and two other AI companies chosen to participate with GE HealthCare presenting during GE’s Innovation
Dr. Randall Jones is one of the speakers this year. Come visit the GE Healthcare Booth #7324 on Nov. 30th at 1:35pm CT.
A prostate cancer diagnosis can be devastating news for the one in seven men who develop the disease in their lifetimes. Annually, 30,000…
We recently sat down with our founder to ask 10 questions about the company, the industry, and what are the options men have when facing…
We recently sat down with our CEO in his library to discuss the mission, vision, and values of Bot Image. We discussed ProstatID and the…
We’re proud to announce that in partnership with our beta testing site, we processed our first test case using the cloud-based architecture on 6/18/2020.
The Bot Image team has been hard at work trying to obtain FDA clearance. We’re excited to announce that we have passed the first major milestone on July 21, 2020.
Trial Site News features the latest coverage of clinical research. Read the article here: